Nitrates and nitrites are compounds that contain nitrogen and oxygen.
Nitrates have three oxygen atoms bonded to the nitrogen, while nitrites have two. Nitrate Nitrates are both the salts and esters of nitric acid. In the biosphere and hydrosphere, nitrates are ubiquitous mainly as sodium nitrate. Autotrophic bacteria convert ammonia to nitrite and then to nitrate under aerobic conditions. When oxygen is not available under anoxic conditions, bacteria can use the oxygen in the nitrate and reduce it to nitrite. Nitrite Nitrites are the salts and esters of nitrous acid.
Nitrite ions are formed by the chemical reaction of nitrous gases with oxygen and moisture in the air (see acid rain) and in the soil in water bodies and in sewage treatment plants by nitrite bacteria (Nitrosomonas) through the oxidation of ammonium ions with the consumption of oxygen. In protein degradation, they are the intermediate product in the complete oxidation of ammonium nitrogen to nitrate (nitrification). They are also formed under anaerobic conditions by bacterial reduction from nitrate ions (nitrate reductase).
HORIBA LAQUAtwin Nitrate Meter
- All Products
-
Parameter
- pH
- ORP
- Conductivity and TDS
- Dissolved oxygen (DO)
- Combined - Multiparameter
- Ammonia (NH3)
- Calcium Ion (Ca2+)
- Chlorid (Cl-)
- Iron (Fe)
- Fluoride (F-)
- Free Chlorine
- Total Chlorine
- Gloss
- Iodine (I)
- Potassium Ion (K+)
- Seawater specific gravity (SSG)
- Sodium Ion (Na+)
- Sodium Chloride (NaCl)
- Nitrate Ion (NO3-)
- Salinity
- Turbidity
- Applications
- Application examples
- Quality
- Brand
- Buffer and calibration solutions
- Spare parts and single components
- Plant analysis - sodium, nitrate, potassium, calcium
- Laboratory Benchtop Meters
- Accessories
Filter products

Mobile Analysis Laboratory for Ion Measurements – Versatile Use Compact kit for simultaneous determination of multiple water and environmental parameters The Horiba 4M LAQUAtwin Kit is a mobile laboratory that allows you to determine important ion parameters directly on-site. The case contains pocket testers for measuring sodium, potassium, nitrate, and calcium, as well as for temperature measurement. Each device uses ion-selective electrodes for targeted analysis of small samples and provides fast, reliable results. In addition to the four measurement devices, the kit includes calibration solutions and a wash/spray bottle for sensor cleaning. The sensors can be stored dry without special storage solutions. Compact Multi-Parameter Kit for Flexible On-Site Analyses The set is designed for measurement tasks where multiple ions need to be detected simultaneously, for example when monitoring drinking and process water, performing environmental checks of water bodies, conducting aquaculture analyses, or agricultural research. Thanks to the case, all essential testers including accessories are always on hand and ready for immediate use. The measurement results are available in a short time and are reliable and reproducible – ideal for routine and control measurements in the field. All components are neatly arranged in the case and ready for use at any time. An additional advantage is that the sensors can be stored dry without special storage solutions. Due to its compact design, the kit is particularly suitable for mobile applications where multiple ion parameters need to be determined quickly one after the other or in parallel. Why Multi-Parameter Ion Measurements Are Useful – and a Mobile Kit Is Ideal In many applications, not just a single measurement is important, but the interaction of multiple ion parameters. Sodium, potassium, nitrate, and calcium together influence water quality, biological processes, plant growth, and technical operations. Single measurements in a laboratory are often time-consuming and inflexible. A mobile analysis kit allows simultaneous detection of multiple parameters directly on-site. This allows relationships to be recognized more quickly, decisions to be made more reliably, and measurement campaigns to be carried out more efficiently. The Horiba 4M LAQUAtwin Kit combines these advantages in a compact, user-friendly system. Additional information We would like to inform you of an upcoming product update for the LAQUAtwin handheld analysers. To enhance product safety and prevent infants and young children from accidentally swallowing the batteries, HORIBA will be introducing a new battery cover on all LAQUAtwin pocket-sized analysers. Overview of the product change Change: Battery cover for enhanced safety. Reason: To prevent accidental swallowing of batteries. Production date: From January 2026 in Japan Deliveries to EMEA distribution partners: From March 2026 As this change is currently being rolled out, mixed deliveries of the current and updated versions may occur during the transition period. Frequently asked questions How long is the warranty period for HORIBA pocket testers? The warranty period is two years from delivery for commercial customers. How many measurements can be taken before the sensor needs to be replaced? This varies depending on the application, use and storage of the Horiba tester. As a rule, the sensor should be replaced after one year. How long is the warranty period for replacement sensors for pocket testers? The warranty period is 12 months after delivery for commercial customers. How do you clean the pocket tester? The pocket tester can be rinsed under water. We recommend cleaning with DI water (deionised). Tissue paper is recommended for drying after cleaning. How can the pocket tester be reset to factory settings? (e.g. in the event of an incorrect measurement result) This information can be found in the operating instructions. These can be found in the download area of the shop and on the product. Is calibration necessary before each measurement? Basically, yes. Further information can be found in the operating instructions.

The High-End LAQUA F-72 forms the top of the laboratory instruments series and measures the parameters pH, ORP, ion, as well as temperature. Ion selective parameters (each with his own electrode): Ammonia ion (NH3), Calcium ion (Ca2+), Chloride ion (Cl-), Fluoride ion (F-), Nitrate Ion (NO3-), Potassium ion (K+) The auto-diagnostic function of the F- series allows the user to ensure that the instruments always provide powerful and reliable measurements. The LAQUA F-72 is fully GLP/GMP compliant and can be connected to an external printer. To ensure traceability, all results are recorded along with sample name, user ID and calibration information. The instrument offers you the proven calibration functions with automatic buffer recognition, stability display as well as the display of the electrode status (wear indicator). We offer a comprehensive range of conductivity electrodes suitable for all types of applications and samples. Known for their stability and durability, HORIBA's electrodes provide accurate measurements under all conditions.LAQUA's large capacitive touch screen panel:Large 5,7" (115,2 x 86,4 mm) graphic touch- color screen Display easy-to-understand menus for values and functionsEffortless single-touch operations- tap, flick and dragSwitch between real-time digital graphs or analogue displays during measurement with a single finger movement Single channel displayExtensive on-screen support on the meter through an electronic user manual with colour images that can be accessed at any time and includes problem-solving instructions for calibration or measurementFeatures:Easy navigation for main instrument and electrode inspections Mode for periodic inspections: JIS/pharmacopoeia/digital simulator Customisable auto-hold function for calibration and measurement Simultaneous connection to a GLP/GMP compliant and a PC Digital memory - up to 2,000 measurement data sets can be recorded360° electrode stand which holds up to three electrodes and is height adjustable USB PC communication and USB memory Multilingual support (Japanese, English, Chinese, Korean)Scope of deliveryF-72 Meterelectrode standprotection coverpower adaptor with 6 plugsdata acquisition software in USB9615S-10D - refillable, glass-body pH electrode with integrated temperature sensor, 1m cable, BNC & phono jack502-S - each 1x pH 4.01, 7.00, 10.01, 3.33M KCl solution (250 ml)

The Dual Channel LAQUA F-73 forms the top of the laboratory instruments series and measures the parameters pH, ORP, ion, as well as temperature. Ion selective parameters (each with his own electrode): Ammonia ion (NH3), Calcium ion (Ca2+), Chloride ion (Cl-), Fluoride ion (F-), Nitrate Ion (NO3-), Potassium ion (K+) The auto-diagnostic function of the F- series allows the user to ensure that the instruments always provide powerful and reliable measurements. The LAQUA F-73 is fully GLP/GMP compliant and can be connected to an external printer. To ensure traceability, all results are recorded along with sample name, user ID and calibration information. The instrument offers you the proven calibration functions with automatic buffer recognition, stability display as well as the display of the electrode status (wear indicator). We offer a comprehensive range of conductivity electrodes suitable for all types of applications and samples. Known for their stability and durability, HORIBA's electrodes provide accurate measurements under all conditions.LAQUA's large capacitive touch screen panel:Large 5,7" (115,2 x 86,4 mm) graphic touch- color screen Display easy-to-understand menus for values and functionsEffortless single-touch operations- tap, flick and dragSwitch between real-time digital graphs or analogue displays during measurement with a single finger movement Switchable single or dual channel displayExtensive on-screen support on the meter through an electronic user manual with colour images that can be accessed at any time and includes problem-solving instructions for calibration or measurementFeatures:Easy navigation for main instrument and electrode inspections Mode for periodic inspections: JIS/pharmacopoeia/digital simulator Customisable auto-hold function for calibration and measurement Simultaneous connection to a GLP/GMP compliant and a PC Digital memory - up to 2,000 measurement data sets can be recorded360° electrode stand which holds up to three electrodes and is height adjustable USB PC communication and USB memory Multilingual support (Japanese, English, Chinese, Korean)Scope of deliveryF-73 Meterelectrode standprotection coverpower adaptor with 6 plugsdata acquisition software in USB Manufacturer article number: 3200575123 (F-73G)

Multifunctional Dual-Channel Laboratory Meter for pH, Redox, Ion-Selective Parameters, and More Professional Horiba F-74 Laboratory Device for Simultaneous Analysis of Multiple Water and Ion Parameters The Horiba F-74 dual-channel laboratory device combines a wide range of analytical measurement methods for various parameters in a powerful system. It measures pH, redox, temperature, conductivity, resistance, salinity, TDS, and ion-selective parameters. The device operates with two independent channels: The first channel is designed for measuring pH, ion-selective parameters, mV, and redox potential. The second channel can measure conductivity, salinity, resistance, and TDS. Automatic buffer and calibration solutions, visual stability indicators, and integrated electrode status ensure reliable and repeatable results. This channel separation allows different parameters to be measured efficiently and simultaneously, supporting safe and convenient operation in the laboratory. Powerful Dual-Channel System for Precise Laboratory Analysis The Horiba F-74 is designed for laboratory environments where measurement accuracy, documentation, and process safety are crucial. Simultaneous recording of multiple parameters saves time and increases the analytical value, especially for complex samples. Its intuitive operation makes the device suitable for both routine analyses and demanding measurement tasks in research, quality control, and industry. Technical Highlights and Key Advantages of the Horiba F-74 Dual-Channel Laboratory Device The laboratory meter offers numerous features that simplify daily use and support compliance with standards: Touchscreen color display for intuitive control and clear visualization Automatic buffer and calibration recognition for reliable results GLP/GMP compliance for regulated applications Data storage and PC communication for easy archiving and analysis The combination of measurement versatility, user-friendliness, and high precision makes the Horiba F-74 an ideal solution for professional multi-parameter laboratory analyses. Frequently asked questions How long is the warranty period for HORIBA analysers? The warranty period is 2 years for the 300 (WQ) series and 1 year for the F series after delivery. Where can I find suitable replacement electrodes or buffer solutions? You can find the appropriate replacement electrodes and buffer solutions in the shop under the main item at the bottom of the cross-selling section. How do I clean the probe before calibration? The sensors should be cleaned with DI water (deionised). After cleaning, wipe them with tissue paper if possible.

Nitrate (NO3-) ion selective electrode for 300 seriesThe new electrode is suitable for the LAQUA WQ-300 series from Horiba and offers precise and reliable measurements for ions in aqueous solutions. It is characterised by its user-friendly handling and environmentally friendly design and can be used for a wide range of applications.Features:Measurement parameters: Nitrate (NO3-) Solid-state electrodes that do not require refillingDouble junction made of porous sintered polyethyleneIntegrated temperature sensor Environmentally friendly design made from bio-based plasticSupplied with calibration and ionic strength adjustment solutionsRobust housing made of polycarbonate and PVCLow maintenance, simply rinse and sealWaterproof according to IP67Linearity in measurement range:The range where the calibration curve has linearity is called the measurement range. When lower concentration is measured where the linearity is not established, perform calibration at two points in the lower concentration range.Influence of pH:The impact of the pH may cause a decrease in the slope of the ion selective electrode, or parallel shifting of the calibration curve. To prevent these effects, set the pH to an appropriate value in advance and keep it constant.Interfering ions:Ion selective electrodes have good selectivity with respect to the ions to be measured, but they may react to the other ions. When using the ion electrode method, it is important to know the effects of coexistent ions and take measures to avoid them. Interfering ions may form compounds with poor solubility or may form complex salts with the structure material of the ion selective membrane, these may to have a significant impact on the measured value.Conditioning:An electrode is conditioned by soaking it in a standard solution for 1 hour or longer. Use a standard solution whose concentration is 100 mg/L or more.In the following cases, perform conditioning of the electrode before calibration.• Before the first use.• Before use after storage for 2 weeks or longer.• If the electrode is less responsive.Calibration:Always calibrate with at least 2 calibration solutions which are 1 decade apart and bracket the expected measurement value.Use fresh calibration solutions and do not re-use them.• Below 10 ppm (mg/L)First solution between 1 and 10ppmSecond solution between 10 and 50 ppm• Between 10 and 100 ppm (mg/L)First solution between 10 and 100ppmSecond solution between 100 and 1000 ppm• Above 100 ppm (mg/L)First solution between 100 and 1000ppmSecond solution above 1000 ppmFurther information can be found in the data sheets in the download area of the store Manufacturer article number: 3200986784 (300-NO3-C)

High-end laboratory measuring instrument for analytics Dear customers, herewith we offer you our highly professional Horiba laboratory table-top measuring instrument of the 2000 series. The LAQUA 2000 series laboratory tabletop meter has a very high-quality 5 inch display with backlighting which shows you all the important information, such as the measured value with up to four decimal places in this version (depending on the resolution), the temperature, the date with time and much more. The LAQUA 2000 series also offers you an adequate device memory with automatic data storage after a set time interval to document your daily measurements accurately over a long period of time. To protect the device settings from unauthorised access, they are password-protected. After your measurements, you can easily transfer all measurement data from the device to your PC via a USB cable in CSV, Excel and Pdf formats using the DAS20 software. In order to facilitate your daily work with the device, the table measuring device visualises the correct calibration, the stable measurement as well as the electrode wear condition. The operation is intuitive because the available keys are marked with symbols and abbreviations. After the analysis, you can clean the device easily and in a time-saving manner with a damp cloth, as the surface is dirt-repellent due to its nature. With the electrode stand included in the scope of delivery, two electrodes can be held and adjusted in height. This allows you to individually align your electrode to your container or glass for calibration and measurement. Special features of the Laqua ION2000 model : The ION2000 is a laboratory meter to which you can connect the separately available ion-selective Horiba probes or determine the pH value and the redox value via the electrodes. Single input, connection of one electrode. Ions (ammonia, calcium, chloride, fluoride, nitrate, potassium) Measurement ion-selective accepts combined and half-cell ion-selective electrodes wide measuring range from 0.000 μg/L to 9999 g/L accepts up to 5 user-defined ion calibration points choice of preferred units: μg/L - mg/L - g/L, ppm - ppt, mmol/L - mol/L Resolution of 4 significant digits after the decimal point with automatic scaling pH-ORP wide measuring range; -2.000 to 20.000 pH high accuracy; ± 0.003 pH resolution: 0.1, 0.01 or 0.001 pH continuous display of calibrated points display of offset and slope - by segment and average up to 5 pH calibration points ± 2000.0 mV 1-point redox calibration automatically recognises and calibrates USA, NIST, NIST2 and DIN pH buffers allows CUST (Custom) buffers for manual calibration Below we would like to show you in full the general functions of the Horiba LAQUA 2000 series: internal memory for up to 2000 data sets, measurement data with time and date information records and stores data in the unit memory based on a specified time interval a 4-digit password protects the meter set-up mode from unauthorised access data can be transferred to a computer and exported in the following formats CSV/Excel/PDF GLP/GMP compliance of the instrument as data can be displayed, recorded and printed with date and time stamp on the instrument Software updates can be downloaded to the instrument Calibration reminder function, indicates that it is time to calibrate (alarm adjustable up to 90 days) Electrode holder is mounted on the meter and can hold up to 2 electrodes (rotates 360º and can be moved up and down 30cm) Displays measurement stability in measurement and calibration mode displays electrode condition based on calibration data smooth surface, easy cleaning RS-232/USB, therefore easy PC and printer connection and free instrument software Ergonomic design for easy viewing and operation Clearly labelled and easy-to-use ports for communication and measurement 5" digital display with backlight, shows all important measurement values and status Automatic calibration, recognises a wide range of standard solutions adjustable switch-off time (up to 30 minutes) temperature compensation, the temperature in °C or °F is either automatically detected if a temperature sensor is connected (ATC mode) or manually entered by the user several measuring modes: Auto-Stable mode signals a stable measured value, Auto-Hold mode holds a stable measured value Real Time mode continuously displays current readings small meter footprint of 155 x 150 mm, saves valuable space Temperature detection, easy display of sample temperature user selectable resolution, up to 3 digits Fixed and individual calibration points Easy navigation, navigate through the settings menu by up, down, right and left Scope of delivery: Measuring device with integrated electrode stand Universal mains adapter with 6 plugs Operating instructions Manufacturer part number: Meter only ION2000 (3200896963)

High-end laboratory measuring instrument for analytics Dear customers, herewith we offer you our highly professional Horiba laboratory table-top measuring instrument of the 2000 series. The LAQUA 2000 series laboratory tabletop meter has a very high-quality 5 inch display with backlighting which shows you all the important information, such as the measured value with up to three decimal places (depending on the resolution), the temperature, the date with time and much more. The LAQUA 2000 series also offers you an adequate device memory with automatic data storage after a set time interval to document your daily measurements accurately over a long period of time. To protect the device settings from unauthorised access, they are password-protected. After your measurements, you can easily transfer all measurement data from the device to your PC via a USB cable in CSV, Excel and Pdf formats using the DAS20 software. In order to facilitate your daily work with the device, the table measuring device visualises the correct calibration, the stable measurement as well as the electrode wear condition. The operation is intuitive because the available keys are marked with symbols and abbreviations. After the analysis, you can clean the device easily and in a time-saving manner with a damp cloth, as the surface is dirt-repellent due to its nature. The electrode stand included in the starter set can be used to hold two electrodes and adjust their height. This allows you to individually align your electrode to your container or glass for calibration and measurement. Special features of the Laqua PC2000 model (multi-parameter): The PC2000 is a combination of the ION2000 and the EC2000 with the advantage that you can measure all parameters with one device. Dual input, connection of 2 electrodes at the same time. pH-ORP wide measuring range; -2.000 to 20.000 pH high accuracy; ± 0.003 pH resolution: 0.1, 0.01 or 0.001 pH continuous display of calibrated points display of offset and slope - by segment and average up to 5 pH calibration points ± 2000.0 mV 1-point redox calibration automatically recognises and calibrates USA, NIST, NIST2 and DIN pH buffers allows CUST (Custom) buffers for manual calibration Conductivity-resistivity-TDS-salinity wide measuring range from 0.001μS/cm to 2,000 S/cm allows automatic and manual conductivity calibrations records individual and average calibration factors after conductivity calibration Calculates resistance, TDS and salinity based on measured conductivity 7 conductivity ranges: 0.000 to 1.999 μS/cm (k = 0.1) 2.00 to 19.99 μS/cm (k = 0.1, 1) 20.0 to 199.9 μS/cm (k = 0.1, 1, 10) 200 to 1999 μS/cm (k = 0.1, 1, 10) 2.00 to 19.99 mS/cm (k = 0.1, 1, 10) 20.0 to 199.9 mS/cm (k = 1, 10) 0.200 to 2.000 S/cm (k = 10) Ions (ammonia, calcium, chloride, fluoride, nitrate, potassium) Measurement ion-selective accepts combined and half-cell ion-selective electrodes wide measuring range from 0.000 μg/L to 9999 g/L accepts up to 5 user-defined ion calibration points choice of preferred units: μg/L - mg/L - g/L, ppm - ppt, mmol/L - mol/L Resolution of 4 significant digits after the decimal point with automatic scaling Below we would like to show you in full the general functions of the Horiba LAQUA 2000 series: internal memory for up to 2000 data sets, measurement data with time and date information records and stores data in the unit memory based on a specified time interval a 4-digit password protects the meter set-up mode from unauthorised access data can be transferred to a computer and exported in the following formats CSV/Excel/PDF GLP/GMP compliance of the instrument as data can be displayed, recorded and printed with date and time stamp on the instrument Software updates can be downloaded to the instrument Calibration reminder function, indicates that it is time to calibrate (alarm adjustable up to 90 days) Electrode holder is mounted on the meter and can hold up to 2 electrodes (rotates 360º and can be moved up and down 30cm). Electrode holder only included in the starter set.Displays measurement stability in measurement and calibration mode displays electrode condition based on calibration data smooth surface, easy cleaning RS-232/USB, therefore easy PC and printer connection and free instrument software Ergonomic design for easy viewing and operation Clearly labelled and easy-to-use ports for communication and measurement 5" digital display with backlight, shows all important measurement values and status Automatic calibration, recognises a wide range of standard solutionsadjustable switch-off time (up to 30 minutes) temperature compensation, the temperature in °C or °F is either automatically detected if a temperature sensor is connected (ATC mode) or manually entered by the user several measuring modes: Auto-Stable mode signals a stable measured value,Auto-Hold mode holds a stable measured valueReal Time mode continuously displays current readings small meter footprint of 155 x 150 mm, saves valuable space Temperature detection, easy display of sample temperature user selectable resolution, up to 3 digits Fixed and individual calibration pointsEasy navigation, navigate through the settings menu by up, down, right and left Scope of delivery: Measuring device with integrated electrode stand Universal mains adapter with 6 plugs Operating instructions Starter set version additionally contains 9625-10D refillable pH electrode with plastic body 9382-10D conductivity electrode with plastic body, cell constant k=1.0 pH 4, 7 and 10 buffer solutions & 3.33M KCl (1 x 250ml each) 84 µS/cm, 1413 µS/cm, 12.88 mS/cm (1 x 250ml each) with electrode holderFlow cell available in combination with 9371-10 stainless steel conductivity cell for ultrapure water measurement (300-2C-C) Manufacturer part number:Meter only PC2000 (3200896964) Starter-set PC2000-SR (1300098882)

Thank you for your interest in our smart and customizable Horiba WQ-300 series instruments.The high-quality digital multi-parameter handheld meters with their generous color LCD display with LED backlight (7 cm x 5.2 cm) are ideally suited for use in the field under all conditions and in direct daylight.Use our configurator to freely select the suitable measuring device of the WQ-300 Horiba series (1 to 3 channels), the measuring parameters (sensor cartridges) and desired cable length (with integrated sensor receptacle + digital module) according to your wishes and requirements. At a later stage, you can always connect another available measurement parameter to the device via the corresponding sensor cable or carry it in the case as a supplement. As with other handheld measuring devices from Horiba, a major advantage of these devices is that the measuring devices are highly resistant to shock, slipping and scratching thanks to a special rubber coating. Only four standard AA 2500 mAh batteries are required for operation, and to give you more flexibility when you are on the move, the WQ-300 series devices can also be powered by a powerbank or battery pack via the integrated USB interface. The IP67 certification, which certifies dustproofness and protection against temporary submersion for up to 30 minutes at a water depth of one meter, completes the overall professional package, along with the usual ergonomic design and ease of user operation. Horiba attests to its outstanding product quality by offering a three-year warranty on the LAQUA WQ-300 series handheld meters. Other added values and features:During analyses, the user is supported by the display of operating instructions in the form of smiley icons, which is supplemented by an LED status display on the sensor. This is intended to prevent operating errors, which can lead to implausible measurement results. The design decision to install only a small number of control buttons and not to implement any nested user menus serves to ensure that the devices can be operated easily and safely even during discontinuous use as well as when used by other users. The measuring instruments have an electrode wear indicator, a measuring stability indicator on the instrument and sensor, a locking function to "save" the currently measured value on the display, free naming of the measuring point (8 characters), automatic calibration functions, integrated sensor holders, self-diagnosis function, as well as GLP functions. The fold-out stand on the back of the instruments means that you always have your hands free to carry out your measurements easily and safely. Measurement data can be conveniently transferred to your computer or cell phone via USB or WiFi.All handheld meters of the 300 series are delivered in a reinforced measuring case for mobile use in the open air. This provides space for the meter, for electrodes and cables, for up to eight 60 ml bottles of buffer solutions including an integrated buffer solution bottle holder for easier calibration. Particularly noteworthy is the automatic sensor recognition of the instrument through the use of sensor cables with integrated digital sensor recording. Information about the sensor, calibration data as well as measurement settings are stored in the sensor recording and transmitted to the measuring instrument. In addition, calibration of all connected sensors is possible at the same time, since each sensor head independently stores the measurement and calibration data and transmits it to the WQ300 series measuring device.Optionally, there is the possibility to connect and use analog sensors via the Horiba BNC sensor adapter with the device, please contact us for this.Languages:GermanEnglishFrenchSpanishPortugueseChineseScope of delivery:WQ-300 series handheld meter in a carrying casethe digital electrode you have chosen, if applicable, and the corresponding sensor-specific cable with integrated digital sensor receptaclemanualManufacturer article number single measuring device WQ-310-K 1 channel: 3200832605 Note: Chloride sensor, if selected, is supplied without chloride ion strength regulator and chloride electrode filling solution. This must be procured by the customer. Without the filling solution the sensor can´t be used. Why We Use These Measuring Devices in Water Treatment Precise calcium control is a key element of efficient water treatment – from drinking water purification and process water management to environmental analysis. Elevated calcium levels can lead to scaling, pipe damage, energy losses, and disruptions in chemical processes. For this purpose, we offer tailored high-end solutions from the Horiba LAQUA WQ Series (WQ310-K, WQ320-K, WQ330-K) in our online shop, specifically designed to meet the needs of water technology: WQ310-K: Single-parameter solution for targeted measurements • WQ320-K: Simultaneous monitoring of, for example, calcium and conductivity • WQ330-K: Three-channel measurement for complex real-time multi-parameter analyses Why We Rely on These Devices: • They provide reliable data for controlling and optimizing water treatment systems • They offer digital evaluation, documented results, and interfaces for process integration • They are robust, packaged in a practical analysis case, and thus also suitable for mobile on-site use • They allow calcium to be measured in combination with other parameters such as pH, conductivity, or redox – for a holistic and efficient approach

Thank you for your interest in our smart and customizable Horiba WQ-300 series instruments.The high-quality digital multi-parameter handheld meters with their generous color LCD display with LED backlight (7 cm x 5.2 cm) are ideally suited for use in the field under all conditions and in direct daylight.Use our configurator to freely select the suitable measuring device of the WQ-300 Horiba series (1 to 3 channels), the measuring parameters (sensor cartridges) and desired cable length (with integrated sensor receptacle + digital module) according to your wishes and requirements. At a later stage, you can always connect another available measurement parameter to the device via the corresponding sensor cable or carry it in the case as a supplement. As with other handheld measuring devices from Horiba, a major advantage of these devices is that the measuring devices are highly resistant to shock, slipping and scratching thanks to a special rubber coating. Only four standard AA 2500 mAh batteries are required for operation, and to give you more flexibility when you are on the move, the WQ-300 series devices can also be powered by a powerbank or battery pack via the integrated USB interface. The IP67 certification, which certifies dustproofness and protection against temporary submersion for up to 30 minutes at a water depth of one meter, completes the overall professional package, along with the usual ergonomic design and ease of user operation. Horiba attests to its outstanding product quality by offering a three-year warranty on the LAQUA WQ-300 series handheld meters. Other added values and features:During analyses, the user is supported by the display of operating instructions in the form of smiley icons, which is supplemented by an LED status display on the sensor. This is intended to prevent operating errors, which can lead to implausible measurement results. The design decision to install only a small number of control buttons and not to implement any nested user menus serves to ensure that the devices can be operated easily and safely even during discontinuous use as well as when used by other users. The measuring instruments have an electrode wear indicator, a measuring stability indicator on the instrument and sensor, a locking function to "save" the currently measured value on the display, free naming of the measuring point (8 characters), automatic calibration functions, integrated sensor holders, self-diagnosis function, as well as GLP functions. The fold-out stand on the back of the instruments means that you always have your hands free to carry out your measurements easily and safely. Measurement data can be conveniently transferred to your computer or cell phone via USB or WiFi.All handheld meters of the 300 series are delivered in a reinforced measuring case for mobile use in the open air. This provides space for the meter, for electrodes and cables, for up to eight 60 ml bottles of buffer solutions including an integrated buffer solution bottle holder for easier calibration. Particularly noteworthy is the automatic sensor recognition of the instrument through the use of sensor cables with integrated digital sensor recording. Information about the sensor, calibration data as well as measurement settings are stored in the sensor recording and transmitted to the measuring instrument. In addition, calibration of all connected sensors is possible at the same time, since each sensor head independently stores the measurement and calibration data and transmits it to the WQ300 series measuring device.Optionally, there is the possibility to connect and use analog sensors via the Horiba BNC sensor adapter with the device, please contact us for this.Languages:GermanEnglishFrenchSpanishPortugueseChineseScope of delivery:WQ-300 series handheld meter in a carrying casethe digital electrode you have chosen, if applicable, and the corresponding sensor-specific cable with integrated digital sensor receptaclemanualManufacturer article number single measuring device WQ-320-K 2 channel: 3200832606 Note: Chloride sensor, if selected, is supplied without chloride ion strength regulator and chloride electrode filling solution. This must be procured by the customer. Without the filling solution the sensor can´t be used.

Horiba WQ330-K – Your Professional 3-Channel Water Quality Meter Expandable multi-parameter system for simultaneous measurement of pH, conductivity, redox, and other water parameters The Horiba LAQUA WQ330-K is a powerful multi-channel device for advanced water analysis, both on-site and in the laboratory. With three freely configurable measurement channels, it can simultaneously record parameters such as pH, conductivity, redox potential, TDS, temperature, oxygen, ammonia, calcium, chloride, nitrate, fluoride, and potassium. Intuitive operation, automatic sensor recognition, and digital calibration ensure accurate, fast, and reproducible measurements. Flexible Multi-Parameter System for Field and Laboratory Applications The analysis kit with the Horiba WQ330-K is ideal for environmental and water body monitoring, research and teaching, as well as drinking and process water control. The large LCD display with LED backlight ensures excellent readability even in direct sunlight. Measurement data can be exported and documented via USB or wirelessly, greatly simplifying traceability and analysis. The modular system allows the device to be quickly and precisely adapted to any measurement task. It is ideal for users who need to efficiently and reliably monitor multiple water parameters. Technical Features and Key Advantages of the WQ330-K The three measurement channels of the Horiba WQ330-K enable simultaneous recording of different water parameters, reducing measurement time and allowing better evaluation of relationships between parameters. The modular sensor concept allows flexible use of digital sensors, which are automatically recognized and store their calibration history. Notably, the combination of high measurement precision, user-friendliness, and mobility makes this device stand out. Quick deployment without complicated setup, along with easy data transfer for documentation and analysis, makes the Horiba LAQUA WQ330-K an ideal solution for professional water analytics. Frequently asked questions How long is the warranty period for HORIBA analysers? The warranty period is 2 years for the 300 (WQ) series and 1 year for the F series after delivery. Where can I find suitable replacement electrodes or buffer solutions? You can find the appropriate replacement electrodes and buffer solutions in the shop under the main item at the bottom of the cross-selling section. How do I clean the probe before calibration? The sensors should be cleaned with DI water (deionised). After cleaning, wipe them with tissue paper if possible.

Precise Nitrate Tester for Plants – Mobile NO₃⁻ Analysis On-Site Horiba Nitrate Ion Pocket Tester Kit for fast and accurate on-site results The Horiba LAQUAtwin Nitrate Ion Hand Tester (NO3-11C) in its practical analysis kit is your mobile laboratory for reliable determination of nitrate ions and temperature in plant sap samples. With a wide measurement range of 6 to 9900 ppm NO₃⁻ and automatic temperature compensation, the device provides fast and accurate results. The ion-selective electrode ensures precise NO₃⁻ measurements, while the digital backlit display allows quick and easy reading of values. Mobile nitrate analysis kit for plants, water, and environmental monitoring The kit contains the handheld tester, five pipettes, three measuring cups, a plant sample press, standard solutions, and sampling sheets. This setup allows for rapid and reproducible analysis of plant sap without the need for additional laboratory equipment. The device is dust- and waterproof, features automatic standard recognition, Auto-Stabil and Auto-Hold functions, and has a long battery life, making it ideal for mobile use in the field, greenhouse, or laboratory. Why nitrate measurement in plants is important – and why a mobile kit is ideal Nitrate content in plants is a key indicator of nutrient supply, plant growth, and agricultural product quality. Excessively high or low nitrate levels can negatively impact yield, plant quality, and environmental compatibility. A mobile nitrate hand tester allows direct analysis of plant sap on-site, without time-consuming laboratory procedures. Fertilization and irrigation measures can be precisely adjusted, and over- or under-supply can be detected early. The Horiba LAQUAtwin NO3-11C provides a precise, fast, and practical solution for this purpose. Typical applications for the Horiba Nitrate Analysis Kit Aquaculture and coral monitoring Aquaristics Horticulture Beverage and food production Laboratories and schools Environmental monitoring Water treatment Water conditioning Additional information We would like to inform you of an upcoming product update for the LAQUAtwin handheld analysers. To enhance product safety and prevent infants and young children from accidentally swallowing the batteries, HORIBA will be introducing a new battery cover on all LAQUAtwin pocket-sized analysers. Overview of the product change Change: Battery cover for enhanced safety. Reason: To prevent accidental swallowing of batteries. Production date: From January 2026 in Japan Deliveries to EMEA distribution partners: From March 2026 As this change is currently being rolled out, mixed deliveries of the current and updated versions may occur during the transition period. Frequently asked questions How long is the warranty period for HORIBA pocket testers? The warranty period is two years from delivery for commercial customers. How many measurements can be taken before the sensor needs to be replaced? This varies depending on the application, use and storage of the Horiba tester. As a rule, the sensor should be replaced after one year. How long is the warranty period for replacement sensors for pocket testers? The warranty period is 12 months after delivery for commercial customers. How do you clean the pocket tester? The pocket tester can be rinsed under water. We recommend cleaning with DI water (deionised). Tissue paper is recommended for drying after cleaning. How can the pocket tester be reset to factory settings? (e.g. in the event of an incorrect measurement result) This information can be found in the operating instructions. These can be found in the download area of the shop and on the product. Is calibration necessary before each measurement? Basically, yes. Further information can be found in the operating instructions.
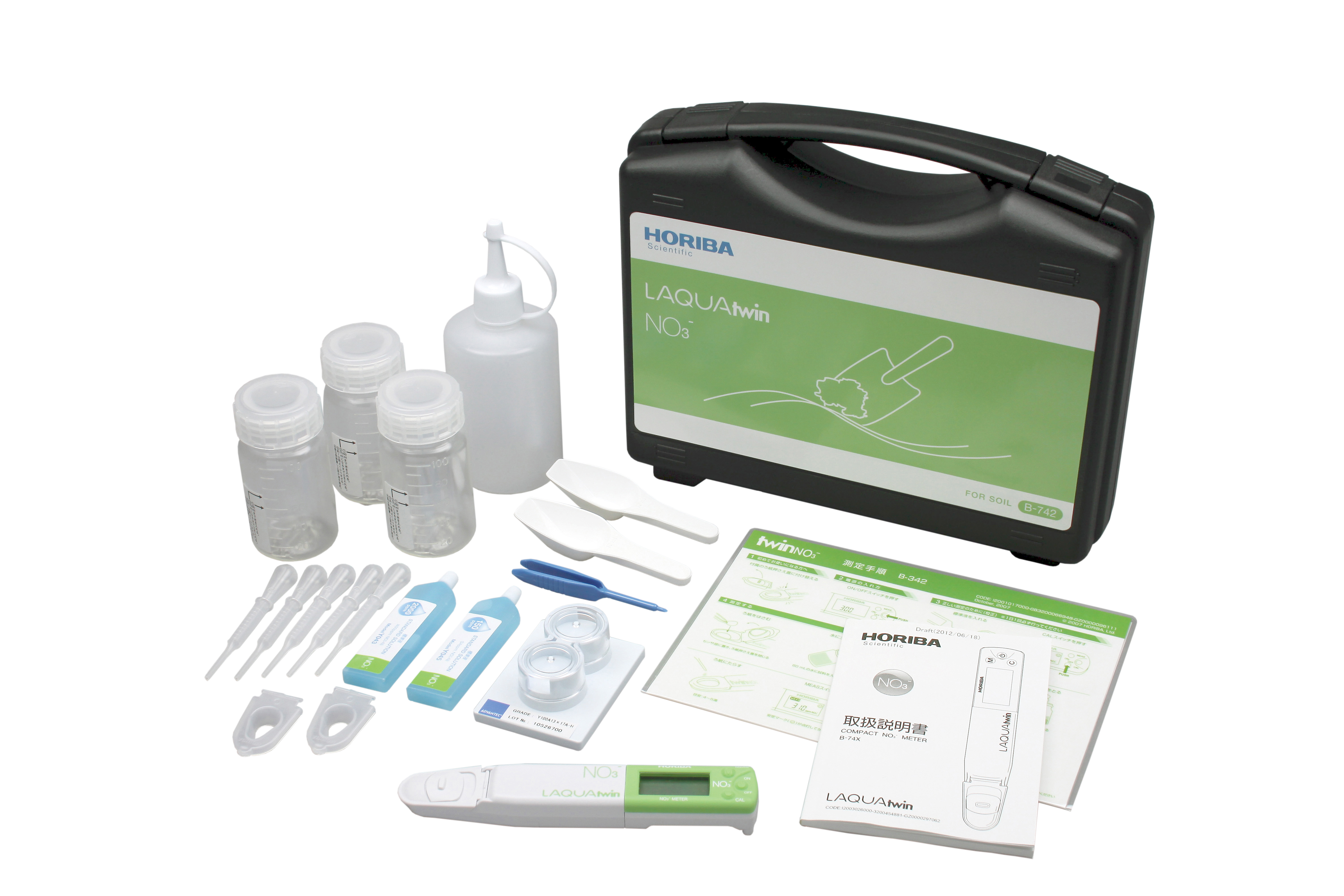
The LAQUAtwin Nitrate Ion Handheld Tester (NO3-11C) included in the analysis kit is pre-set by the factory to the relevant calibration points 30PPM and 300PPM for soil analysis. Manufacturer Part No. 3200689164 | B-742 Additional information We would like to inform you of an upcoming product update for the LAQUAtwin handheld analysers. To enhance product safety and prevent infants and young children from accidentally swallowing the batteries, HORIBA will be introducing a new battery cover on all LAQUAtwin pocket-sized analysers. Overview of the product change Change: Battery cover for enhanced safety. Reason: To prevent accidental swallowing of batteries. Production date: From January 2026 in Japan Deliveries to EMEA distribution partners: From March 2026 As this change is currently being rolled out, mixed deliveries of the current and updated versions may occur during the transition period.

Horiba LAQUAtwin Nitrate Ion Tester NO3-11 for Precise On-Site Measurements Handheld tester with 2-point calibration, ideal for water and environmental measurements The Horiba LAQUAtwin Nitrate Ion Tester (NO3-11) is a portable device for direct determination of nitrate ions (NO3) and temperature in liquids and samples. Thanks to the ion-selective electrode and compact design, the tester delivers fast and reliable results. The integrated temperature unit enables temperature-compensated analysis and clearly displays both nitrate and temperature values on the digital LCD display. Mobile Nitrate Ion Tester for Precise On-Site Analyses The integrated temperature measurement enables temperature-compensated analysis and shows both nitrate and temperature values. Only very small sample volumes are required, minimizing sample consumption and increasing efficiency in daily use. The tester can be set up with up to two calibration points, with features such as automatic standard recognition, adjustable calibration values, and multiplication compensation for flexibility. Only minimal sample volumes are needed for each analysis, reducing sample usage and improving efficiency. Why Nitrate Measurement Is Crucial – and a Pocket Tester from Horiba Is Ideal Nitrate measurement plays a central role in agriculture, water management, and environmental monitoring. Elevated nitrate concentrations can affect drinking water quality, stress ecosystems, and contribute to over-fertilization. At the same time, nitrate is an essential nutrient for plants, and targeted control is vital for sustainable agriculture. A mobile nitrate pocket tester enables rapid on-site analysis without time-consuming laboratory tests. This allows early detection of contamination, immediate action, and reliable monitoring of legal limits. The Horiba LAQUAtwin NO3-11 combines this flexibility with high measurement accuracy and easy operation – ideal for routine checks and scientific applications. The tester can be configured with up to two calibration points. Features like automatic standard recognition, adjustable calibration values, and multiplication compensation provide high adaptability to different sample types and measurement conditions. Typical Applications of the Horiba LAQUAtwin Nitrate Ion Tester (NO3-11) Agriculture: To optimize fertilizer use, promote plant growth, and minimize environmental impact, farmers and agronomists use the tester to monitor nitrate levels in soils and irrigation water. Water Quality Management: To maintain nitrate concentrations within safe limits, the NO3-11 is used in water management to check drinking and wastewater quality. Environmental Monitoring: The NO3-11 allows environmental technicians and scientists to quickly analyze nitrate contamination in rivers, lakes, and groundwater, ensuring compliance with environmental standards and identifying pollution sources. Research and Development: The tester is used by scientists in environmental science, hydrology, and soil science to conduct detailed studies of the nitrogen cycle and the effects of nitrates on ecosystems. Additional information We would like to inform you of an upcoming product update for the LAQUAtwin handheld analysers. To enhance product safety and prevent infants and young children from accidentally swallowing the batteries, HORIBA will be introducing a new battery cover on all LAQUAtwin pocket-sized analysers. Overview of the product change Change: Battery cover for enhanced safety. Reason: To prevent accidental swallowing of batteries. Production date: From January 2026 in Japan Deliveries to EMEA distribution partners: From March 2026 As this change is currently being rolled out, mixed deliveries of the current and updated versions may occur during the transition period. Frequently asked questions How long is the warranty period for HORIBA pocket testers? The warranty period is two years from delivery for commercial customers. How many measurements can be taken before the sensor needs to be replaced? This varies depending on the application, use and storage of the Horiba tester. As a rule, the sensor should be replaced after one year. How long is the warranty period for replacement sensors for pocket testers? The warranty period is 12 months after delivery for commercial customers. How do you clean the pocket tester? The pocket tester can be rinsed under water. We recommend cleaning with DI water (deionised). Tissue paper is recommended for drying after cleaning. How can the pocket tester be reset to factory settings? (e.g. in the event of an incorrect measurement result) This information can be found in the operating instructions. These can be found in the download area of the shop and on the product. Is calibration necessary before each measurement? Basically, yes. Further information can be found in the operating instructions.
